Rare Earth Elements Project
The award-winning Rare Earth Elements Project explored the historical and contemporary challenges posed by the rare earth metals. These metals are central to electronics, green energy, medical technologies, and more. Chemists have long struggled to separate them efficiently, while often producing a lot of pollution. China controls most production today, making the rare earths a flashpoint in international relations.
The project’s capstone program—“Rare Earth Elements: The Intersection of Science and Society”—was held in Los Angeles on September 24, 2019. Led by award-winning science correspondent and Science Friday host Ira Flatow, this event brought together scientists, journalists, and the scientifically curious to consider the potentials and challenges of extracting and refining rare earth minerals.
The Rare Earth Elements Project also included a role-playing game for students, a survey of Americans’ rare earth knowledge, and a two-part podcast episode about the costs of producing rare earths. We continue to explore these critical metals and to develop new content (see below for links).
About Rare Earth Elements
Despite their name, rare earth metals are not rare at all. Their unique magnetic, luminescent, and electrochemical properties make them essential to such products as smartphones, electric cars, camera lenses, X-ray scanners, wind turbines, and computer hard drives.
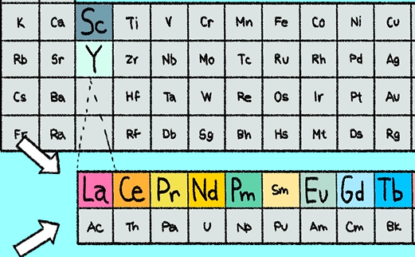
There are 17 rare earth elements on the periodic table: scandium (Sc), yttrium (Y), lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), and lutetium (Lu).

DISTILLATIONS PODCAST
Rare Earths: The Hidden Cost to Their Magic
Rare earth elements are essential to modern life. Luckily the world has plenty of them; unfortunately, getting them out of the ground leaves behind massive environmental damage.

VIDEO
Rare Earth Elements: The Intersection of Science and Society
Led by noted science journalist Ira Flatow, this panel discussion featured leading researchers sharing their insights into these increasingly significant elements.

DISTILLATIONS MAGAZINE
Choosing a Better High-Tech Future
Rare earth elements make modern devices faster, brighter, and lighter, but it will take the creaky gears of government to make their production cleaner and more equitable.

EDUCATION
The Case of Rare Earth Elements
Part of our Conflicts in Chemistry role-playing game series, students debate the positive and negative perspectives of rare earth elements from all sides.
![Europium (III) Hydroxide [Eu(OH)₃] under UV Light](https://www.sciencehistory.org/wp-content/uploads/2023/04/europium_iii_hydroxide_under_uv_light-415x257.jpg)
DIGITAL EXHIBITIONS
Critical Metals: The Chemistry of Light
Turn on the illuminating history and science of rare earth metals.
EDUCATION
Rare Earth Elements and Why They Matter
The importance of understanding the elements of technology.

EXHIBITIONS
Enabling Transparency
Glass artist Anna Mlasowsky examines the history of the rare earth elements and their socio-ecological impact on local communities and the environment.

DIGITAL COLLECTIONS
Rare Earths Collection
This digital collection features materials related to rare earth elements.

DISTILLATIONS MAGAZINE
Industrial Vitamins
Rare earth metals are the vitamins of modern technology. How did this group of chemically dull elements become so important and so troublesome?
The Rare Earth Elements Project was made possible by a generous grant from Roy Eddleman, founder of Spectrum LifeSciences.
